NB Naturalist Feature: Newly Recorded Tapeworms Infecting Fishes of New Brunswick and the Need for Citizen Science!
By Megan Fraser
Parasites are organisms that live on or inside of a host organism; the host suffers some level of harm and the parasite benefits (Loker & Hofkin 2015). Parasitism is a common symbiotic lifestyle, and parasites are present in every kingdom of life in many shapes and sizes. Almost all plants and animals are hosts to one or more parasitic organisms. The study of parasites and their hosts is known as parasitology, and the study of parasitic worms specifically is termed helminthology. Parasitic worms belong to a group of parasites called “helminths”, which includes classes such as the nematodes (roundworms), trematodes (flukes), and cestodes (tapeworms). Helminths can be ectoparasites which are specialized to live on the outside of their host, or endoparasites which live inside of their host. To the average person the word “tapeworm” evokes many emotions, mostly those of disgust and repulsion. As odd and admittedly disturbing as tapeworms are, they are fascinating, nonetheless. Not only do tapeworms rob their host of nutrients; some species also modulate their host’s behavior, immune system, and reproductive fitness. Fish are no strangers to helminth infections, with an estimated 30,000 helminth species infecting fish worldwide (Jyrwa et al. 2016)!
I was given the opportunity to dive into the field of helminthology after several tapeworms retrieved from fish in New Brunswick were sent to Parasitologist Dr. Michael Duffy (UNBF) and passed along to me for identification. Species level identification of tapeworms is difficult, especially during their larval stages. Tapeworms have few distinctive morphological features, and some species infect multiple host fish, so host identity cannot be used for specific cestode identification. Species level identification is further complicated by the fact that many tapeworms are yet to be discovered and named!


Tapeworms were recently collected from fish from two separate locations in New Brunswick (Fraser et al. 2023). The first set was retrieved in 2017 from Slimy sculpin in northern New Brunswick, during unrelated investigations on the impacts of forest management on fish health (White 2020, Negrazis et al. 2022). The second set was collected in 2021, following liberation from their host during a fish survey in southern New Brunswick. More of these tapeworms were retrieved from Blacknose dace at the same location in 2022. Upon comparison of the northern and southern parasite specimens two morphological features were obvious; the worms from northern New Brunswick were segmented and relatively short (Figure 1); and the worms from southern New Brunswick were unsegmented and relatively long (Figure 2). Given these morphological differences, I knew that I was dealing with two separate parasite species. I proceeded by collecting a small piece from the tip of each worm and extracting DNA. Upon comparing the DNA to that of all the known tapeworms available in a large genetic sequence database (GenBank), I made two discoveries:
1. The worms from northern New Brunswick have 89-92% identity with tapeworms of the Schistocephalus genus, confirming that these worms belong to the Schistocephalus genus, and are potentially a new species.
2. The worms from southern New Brunswick have >99% identity with the tapeworm Ligula intestinalis, which enabled definitive species identification of this parasite. Ligula intestinalis had not been recorded previously from New Brunswick.
Schistocephalus species are typically regarded as specialists to the Threespine and Ninespine stickleback, but the worms from northern New Brunswick were found in Slimy sculpin. It appears that an undocumented host switch and speciation event may have occurred in the Schistocephalus genus, giving rise to the worms found in the Slimy sculpin from northern New Brunswick. This is potentially a new species, and a very interesting scientific discovery!
The lack of previous records of L. intestinalis in New Brunswick is noteworthy. Our recent identification raises the question of whether this tapeworm was recently introduced, or if it has been present for many years without being identified. Ligula intestinalis has three hosts during its life cycle (Arme & Owen 1968, Szalai et al. 1989, Loot et al. 2002, Biswas & Ash 2021). It reproduces in the intestine of a fish-eating bird and eggs pass with the bird’s faeces into a body of freshwater. Eggs hatch before being eaten by tiny crustaceans known as “copepods”; infected copepods are eaten by fish, infected fish are eaten by birds, and the lifecycle repeats. Ligula intestinalis worms can become very large, growing up to 100 cm in length (Yoneva et al. 2015)! Recent introduction of L. intestinalis to New Brunswick is possible, especially given the highly mobile nature of the bird host. However, it is also possible that L. intestinalis had not been identified from New Brunswick due to a lack of research focus on cestodes. This question remains to be answered.
Ligula intestinalis can “parasitically castrate” its host fish. In other words, the infection severely limits the ability of infected fish to reproduce. This castration effect is reported in many European fish species including chub, roach, rudd, bream, minnow, and dace; however, this is not well studied in North American fish populations. Infected fish can present with little-to-no gonadal growth, less prominent spawning tubercles, significantly lower levels of sex hormones, and may exhibit abnormal behavior during their spawning period (Orr 1966, Arme & Owen 1968, Carter et al. 2005, Hecker & Karbe 2005, Schabuss 2005, Trubiroha et al. 2009, Trubiroha et al. 2010). The mechanism that causes castration of the fish host is unknown, however it is speculated to be a protein or other molecule actively secreted by the parasite! In the laboratory, my next steps will involve collecting secretions from these tapeworms and identifying the major secreted molecules. Identifying these molecules will hopefully bring us a step closer to better understanding the mechanisms that lead to parasitic castration of their fish host.
I aim to determine the geographic range of L. intestinalis by monitoring natural fish populations in New Brunswick; is this parasite restricted to southern New Brunswick lakes, or does it occur throughout the province? Knowing the geographic range is important given the potential negative implications of L. intestinalis infections on host reproduction, especially in threatened fish populations. This is where I decided that a citizen science initiative would be helpful! Recreational fishermen often find tapeworms in wild caught fish. By engaging the public, I hope to have recreational fishers retain tapeworms that they find so that I may collect them for identification. If any specimens match with L. intestinalis, this will give us valuable insight in terms of the parasite’s range across New Brunswick, and the species of fish that can act as hosts. Please contact Megan Fraser (email: mfrase12@unb.ca) if you find tapeworms while fishing, and specimen pick-up will be arranged!
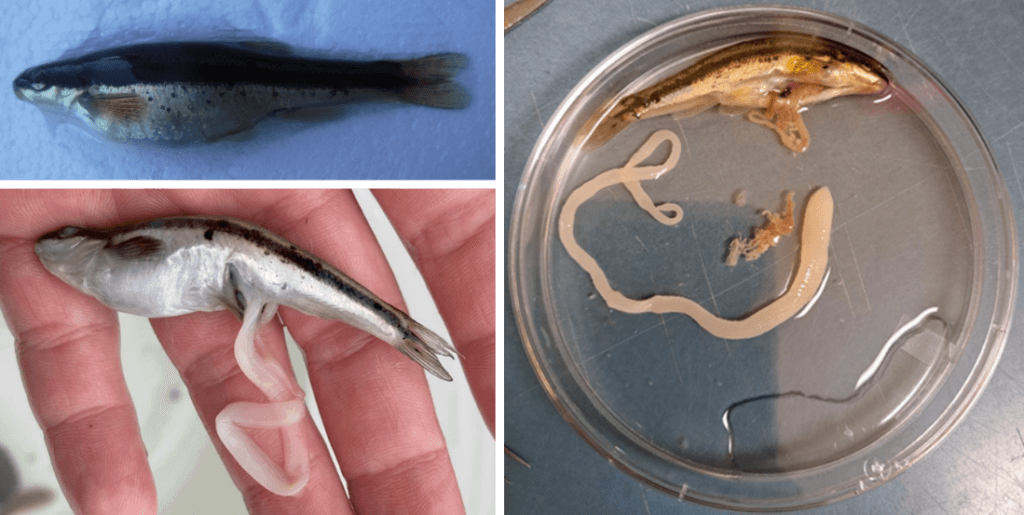
Our use of collections from recreational fishermen will reduce the need to sacrifice additional fish for our own surveys. During Spring-Fall of 2023 I will perform a survey of several lakes across New Brunswick to answer questions regarding parasite range, permissive fish hosts, and seasonal trends in the prevalence of infection. Morphological indicators of infection such as abdominal swelling or tapeworms erupting from the vent (the opening where eggs, sperm, and waste leave the fish) will help us to select fish to sample (Figure 3). To diagnose a fish with this infection, past methodologies required euthanasia and dissection. We will be modifying a non-lethal, minimally invasive diagnostic procedure from Berger and Aubin-Horth (2018) to allow us to diagnose infected fish without euthanasia. Our goal is to collect samples from recreational fishermen and use non-lethal diagnostic techniques, to accomplish our survey with the least possible ecological disturbance. Our studies will contribute valuable information to the field of parasitology in New Brunswick, and to parasitological work involving Ligula intestinalis worldwide!
Acknowledgements:
I thank Dr. Michelle Gray, Ben Andrews, and Sarah Van De Reep for collecting the original tapeworms for identification. Without their interest and curiosity, I would not be involved in this important research project. I thank my MSc Supervisory Committee, Dr. Michael Duffy, Dr. Shawn MacLellan, and Dr. Michelle Gray for their advice and guidance. I also thank Kerstyn Dobbs, Hannah Lazaris-Decken, Nathanael Moore, Stephanie Scott, and Tyler Lynn from UNBF for their assistance in the laboratory; and Sarah Van De Reep, Becky Graham, Sara Plant, Allain Cassie, and John Robinson from Parks Canada for their assistance in the field.
References:
- Arme, C., & Owen, R. W. 1968. Occurrence and pathology of Ligula intestinalis infections in British fishes. The Journal of Parasitology, 54, 272–280.
- Berger, C. S., & Aubin-Horth, N. 2018. An eDNA-qPCR assay to detect the presence of the parasite Schistocephalus solidus inside its Threespine stickleback host. Journal of Experimental Biology, 221(9), jeb178137.
- Biswas, R., & Ash, A. 2021. Impact of Endocrine Disruption on Host-Parasite Interaction: A Digest from a Cosmopolitan Cestode Model Ligula intestinalis. In Proceedings of the Zoological Society. pp. 1-9. Springer India.
- Carter, V., Pierce, R., Dufour, S., Arme, C., & Hoole, D. 2005. The tapeworm Ligula intestinalis (Cestoda: Pseudophyllidea) inhibits LH expression and puberty in its teleost host, Rutilus rutilus. Reproduction, 130, 939-945.
- Fraser, M.L., M.A. Gray, K.D.R. Dobbs, B.I. Andrews, S. Van De Reep, M. S. Duffy. 2023. First reports of Ligula intestinalis and a Schistocephalus sp. infecting small-bodied fish in New Brunswick, Canada. Journal of Parasitology 109:288-295. DOI: 10.1645/22-127
- Hecker, M., & Karbe, L. 2005. Parasitism in fish—an endocrine modulator of ecological relevance?. Aquatic Toxicology, 72, 195-207.
- Jyrwa, D. B., Thapa, S., & Tandon, V. (2016). Helminth parasite spectrum of fishes in Meghalaya, Northeast India: a checklist. Journal of Parasitic Diseases, 40, 312-329.
- Loker, E., & Hofkin, B. 2015. Parasitology: a conceptual approach. Garland Science, Taylor & Francis Group, LLC. 711 Third Avenue, New York, NY, 10017, USA
- Loot, G., Aulagnier, S., Lek, S., Thomas, F., & Guégan, J. F. 2002. Experimental demonstration of a behavioural modification in a cyprinid fish, Rutilus rutilus (L.), induced by a parasite, Ligula intestinalis (L.). Canadian Journal of Zoology, 80, 738-744.
- Negrazis, L., Kidd, K. A., Erdozain, M., Emilson, E. J., Mitchell, C. P., & Gray, M. A. 2022. Effects of forest management on mercury bioaccumulation and biomagnification along the river continuum. Environmental Pollution, 310, 119810.
- Orr, T. S. C. 1966. Spawning behaviour of rudd, Scardinius erythrophthalmus infested with plerocercoids of Ligula intestinalis. Nature, 212, 736-736.
- Schabuss, M., Gemeiner, M., Gleib, A., Lewis, J. W., Miller, I., Möstl, E., & Grillitsch, B. 2005. Ligula intestinalis infection as a potential source of bias in the bioindication of endocrine disruption in the European chub Leuciscus cephalus. Journal of Helminthology, 79, 91-94.
- Szalai, A. J., Yang, X., & Dick, T. A. 1989. Changes in numbers and growth of Ligula intestinalis in the spottail shiner (Notropis hudsonius), and their roles in transmission. The Journal of Parasitology, 75, 571–576
- Trubiroha, A., Kroupova, H., Wuertz, S., Frank, S. N., Sures, B., & Kloas, W. 2010. Naturally-induced endocrine disruption by the parasite Ligula intestinalis (Cestoda) in roach (Rutilus rutilus). General and Comparative Endocrinology, 166, 234-240.
- Trubiroha, A., Wuertz, S., Frank, S. N., Sures, B., & Kloas, W. 2009. Expression of gonadotropin subunits in roach (Rutilus rutilus, Cyprinidae) infected with plerocercoids of the tapeworm Ligula intestinalis (Cestoda). International Journal for Parasitology, 39, 1465-1473.
- White, C. F. H. 2020. Fish effects from forest harvest and ectoparasitic copepods in northern New Brunswick. MScF Thesis, University of New Brunswick, 120 pp.
- Yoneva, A., Scholz, T., M?ocicki, D., & Kuchta, R. 2015. Ultrastructural study of vitellogenesis of Ligula intestinalis (Diphyllobothriidea) reveals the presence of cytoplasmic-like cell death in cestodes. Frontiers in zoology, 12, 1-9.




